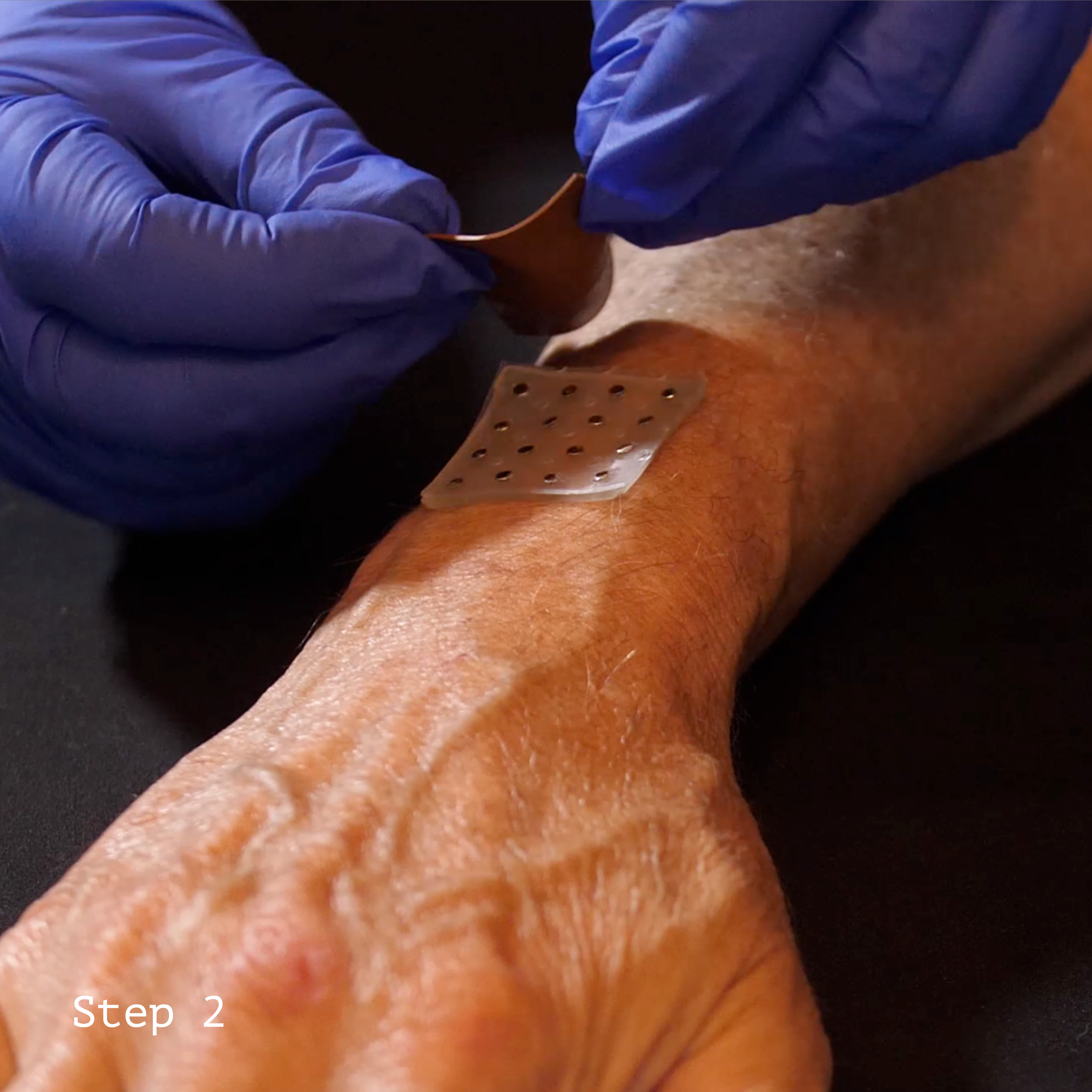
Example of CivaDerm™ Usage
CivaTech Oncology’s new device, CivaDerm has received FDA clearance. CivaDerm is a temporary radiation therapy product designed for intraoperative or surface radiation to treat skin cancer and other lesions.
- Cleared by the FDA for intraoperative and surface radiation
- Starting 30 patient clinical study to determine outcomes for skin cancer over 6 weeks
- To be used in bandage to treat skin disease in 5 days, eliminating surgery for non-melanoma skin cancers, keloids and other lesions that respond to radiotherapy
- Better treated with a single application followed by a single removal
- No expensive capital equipment purchase required by treatment facilities
- Patients do not need to make repeated visits to the treatment facility during the course of therapy
- Excellent treatment options for elderly patients who may not tolerate surgery and/or have difficulty commuting for repeated radiation treatment visits
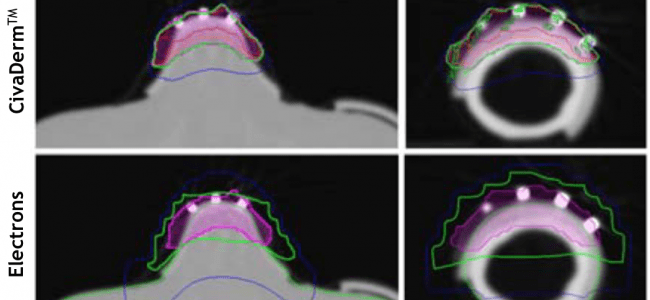
CivaDerm radiation dosimetry plans have been compared to linac-based electron therapy plans. Multiple areas of the face and body were evaluated. Two comparison dose distributions for the nose and for the finger are shown in the figure below. “100% isodose line for [CivaDerm™] plans more closely correlated with the treatment depth than electron plans, and were more conformal to the curved surfaces.” Additionally, the skin surface dose was found to be acceptable for the CivaDerm™ plans.
CivaDerm with a 3mm bolus can “provide equivalent or better dose distribution than electron therapy.”
CivaDerm is an alternative to electron therapy for areas that are:
- Difficult to set up on a linear accelerator
- Curved
- Better treated with a single application followed by a single removal
1