Innovating Localized Radiation Therapy
Pancreatic cancer survivor discusses CivaSheet® Implant

CivaTech Oncology® designs innovative products that bring meaningful improvements to low dose rate brachytherapy. The CivaString® and CivaSheet® can substantially reduce procedure time, provide homogeneous dose distribution and minimize trauma. Customizable CivaTech products insure that radiation oncologists can deliver individualized, targeted therapy to treat cancer.
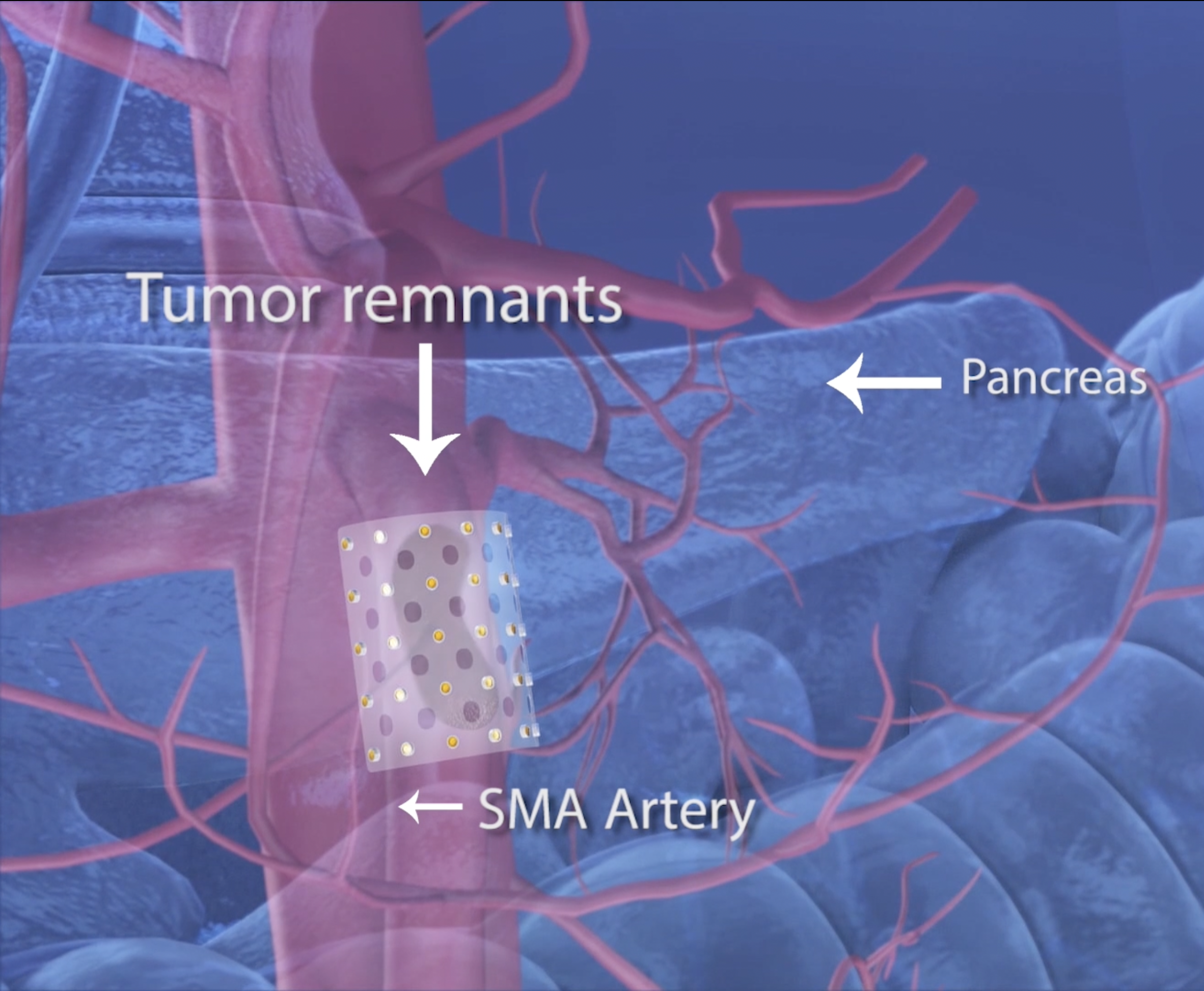
CivaSheet®
CivaSheet® is the only FDA cleared, unidirectional, planar brachytherapy source. It is flexible, bioabsorbable and features CivaTech’s proprietary polymer encapsulated source technology.
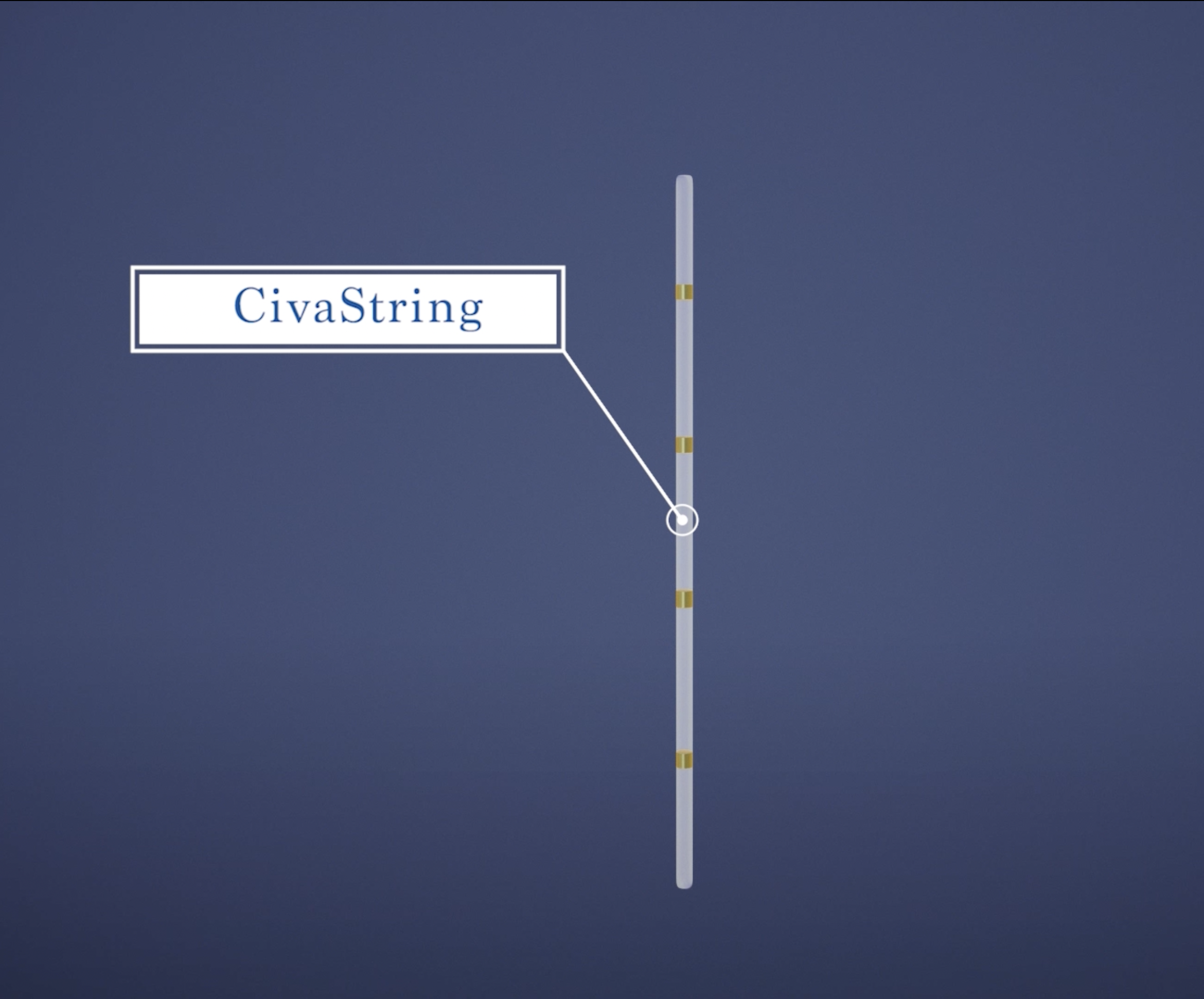
CivaString®
CivaString® is a unique linear, polymer-encapsulated, low-dose-rate (LDR) brachytherapy source used to treat localized, solid tumors.

CivaDerm™
CivaDerm™ is a temporary radiation therapy product designed for intraoperative or surface radiation to treat skin cancer and other lesions.
Polymer source technology… What are the advantages you should know?
- Only homogenous sources – eliminates hot/cold spaces
- No source migration in clinical studies
- Eliminates potential of long term metal toxicities
The novel CivaString®, CivaSheet® and CivaDerm™ seamlessly integrate into current clinical workflows, beginning with your usual imaging modalities, through treatment planning, and ending with post-implant dosimetry.
“Dr. Prestidge indicated he got 99.9% coverage of the prostate using 12 needles. I perhaps was in recovery for less than an hour. I felt very very comfortable; I had virtually no discomfort for a good period of time.”
“I got the CivaString and the results have been excellent! My first PSA after implant was .35 and my second PSA was .20. I am scheduled for my third blood test in July. Your product seems to have done the trick. I’m 75 and back playing racquetball”
“Excellent quality implants across a range of gland sizes were accomplished. Prostate volume changes were minimal suggesting minimal swelling using the CivaString needles and strands. The product is robust with minimal movement (4%) evidenced by CT. Good dosimetry is achieved with both the pre-plans and post-plans. Using a line source, the number of needles required to deliver the radiation therapy is less than that reported with standard preloaded or stranded brachytherapy products. This study demonstrates that the CivaString product can be safely and effectively integrated into the LDR brachytherapy management of prostate cancers.”
Notable Achievements
-
CivaSheet® is the only bioabsorbable and directional radiation therapy device approved by the FDA.
-
Manufacturing meets FDA guidelines for medical devices and highest international standards of ISO 13485 for production in a clean room environment.
-
The Company has two licenses with the NRC Agreement State of North Carolina. One license allows CivaTech to conduct broad research and the other allows for the manufacture and distribution of products containing radioactive material.
-
The CivaSheet® has been supported by the NIH/NCI
-
CivaString® and CivaSheet® have both been FDA cleared to treat cancers of any solid tumor such as Prostate, Pancreas, Sarcoma, Head and Neck, Gyn, Colorectal, Breast, Brain, etc.
About Us
CivaTech Oncology Inc. is a North Carolina-based corporation that offers cancer patients radiation therapy treatment options designed to minimize distress, maximize quality of life, and improve life expectancy. The company has developed and is commercializing innovative polymer-based low dose rate brachytherapy devices, setting a new standard of care in the treatment of various stages of cancer.